More Information
Submitted: February 24, 2026 | Accepted: March 05, 2026 | Published: March 06, 2026
Citation: Mangano S, Lopatriello S. Experience of Patients with Vascular Access for Hemodialysis VALE Project-Vascular Access Life Experience. J Clini Nephrol. 2026; 10(3): 021-031. Available from:
https://dx.doi.org/10.29328/journal.jcn.1001172
DOI: 10.29328/journal.jcn.1001172
Copyright license: © 2026 Mangano S, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Vascular access; Hemodialysis; Qualitative analysis; Patient engagement
Experience of Patients with Vascular Access for Hemodialysis VALE Project-Vascular Access Life Experience
Stefano Mangano and Stefania Lopatriello*
Department of Nephrology, Tradate Hospital, Varese; Kint-sugi, Via Marciano di Eraclea, Rome, Italy
*Corresponding author: Stefania Lopatriello, MPharm, Kint-sugi, Via Marciano di Eraclea, Rome, Italy, Email: [email protected]
Rationale & objective: For individuals with end-stage kidney disease (ESKD) undergoing hemodialysis (HD), vascular access (VA) is fundamental for extending their lives and enhancing their quality of life (QoL). Placing patients at the heart of dialysis access decisions is crucial, not just addressing immediate access requirements but also looking ahead to their future needs. To facilitate better, it’s essential to understand how various VA methods affect the patient’s overall experience.
Study design: Patient experiences with arteriovenous fistulas (AVFs), arteriovenous grafts (AVGs), and central venous catheters (CVCs) were investigated through individual, semi-structured phone interviews, which utilized a validated questionnaire as a framework.
Setting & participants: Adult ESKD patients undergoing HD for at least six months, including those with prior kidney transplants, were selected. Participants were over 18, ANED (National Association of Dialyzed and Transplanted) members, and volunteers.
Analytical approach: The study’s theoretical framework was built using grounded theory principles. Initial coding was based on themes from a validated questionnaire (Nordyke, et al. 2020), and further codes and subcodes were generated through inductive coding (MAXQDA-2022 software).
Results: The study found significant variation in VA types: 30.0% (N = 30) AVF only, 26.7% CVC to AVF, 13.3% AVF to CVC, 10.0% graft, 6.7% stable VA history, and 13.3% complex histories. Appropriate CVC discontinuation was low (10%). VA type significantly impacted social and daily activities/physical functioning, symptoms, and sleep. Younger patients (≤65%) experienced reduced QoL in work, family, and leisure. Reduced QoL stemmed from impaired daily activities, attention deficits, fear, anxiety, and worry. AVF had a greater impact than CVC and AVG in almost all domains. Patients preferred fistulas, regardless of HD years and geographical location.
Limitations: The absence of inclusion of patients from surgical hubs may not represent the real-world variety of Italian dialysis center VA protocols.
Conclusion: Healthcare providers should discuss VA options and their impact on QoL, considering patients’ life goals and preferences.
The chronic care model reported in the 2024-KDIGO (Kidney Disease: Improving Global Outcomes) guidelines [1] emphasizes, among factors of improvement of end-stage kidney disease (ESKD) outcomes, the presence of informed, activated patients and their performing productive interactions with a prepared, proactive multidisciplinary team. Besides, the 2019 Kidney Disease Outcomes Quality Initiative guidelines (2019-K-DOQI guidelines) [2] recommend that “each patient with progressive CKD … should have an individualized ESKD Life-Plan that is regularly reviewed, updated, and documented on their medical record”. The ESKD Life-Plan is a collaborative strategy between patients and a multidisciplinary team to manage ESKD. It begins before dialysis is needed and covers the entire journey from chronic kidney disease to ESKD. The plan considers individual needs and goals to optimize treatment choices and dialysis access. It’s a dynamic process, adapting to changes in the patient’s life and medical conditions. A patient-centered approach to dialysis access is critical, considering not only current access needs but also proactively planning for the next one. This approach goes beyond simply creating and maintaining access; it’s about tailoring a long-term strategy that aligns with the patient’s individual needs, preferences, and circumstances. This holistic, forward-thinking strategy is the core of the ESKD Life-Plan [2].
Vascular accesses (VA) are considered a lifeline by patients with ESKD. Diverse types of VAs are used to facilitate hemodialysis (HD) in patients with ESKD: arteriovenous fistulas (AVF), arteriovenous grafts (AVG), and central venous catheters (CVC). Selecting the “best” type of access is complex and requires a multidisciplinary approach, considering patient preferences and values, improving traceability of outcomes, and conducting cyclical reassessments. Patients often feel uninvolved in this process and are poorly informed about the risks and benefits of each type of VA [3]. A recent review showed that, although patient experience is very relevant, patient-reported outcomes are rarely used in the decision-making process [4].
In this framework, to assess real-world, patient-reported outcomes of patients with HD due to ESKD. A qualitative investigation was performed on a pool of Italian HD patients with ESKD and aimed at understanding their psychosocial experience, including physical function, emotional impact, family and social relationships, ability to work or attend school, sleep patterns, and interactions with HD centers. The research goals also included the time until HD access (“delay”).
Study design
Semi-structured one-to-one interviews were conducted via phone. The study followed the Consolidated Criteria for Reporting Qualitative Research reporting guideline [5]. Consent to participate was collected from patients based on the EU GDPR laws n. 2016/679 as participants were associated with ANED (Associazione Nazionale Emodializzati Dialisi e Trapianto).
Setting and participants
The patients were contacted and invited to participate by ANED based on a screener flowchart defined by the Authors. Inclusion criteria were: patients with ESKD, age ≥18 years, duration of HD of at least 6 months, any type of VA used, regardless of prior kidney transplantation. If interested in the study, they were required to sign the informed consent. Exclusion criteria were defined if one of the flowchart steps was denied.
Subsequently, the consecutively selected patients were invited to complete a semi-structured phone interview while maintaining anonymity.
Interview guide
The semi-structured interview was developed from Nordyke, et al. 2020 [2], a validated questionnaire on the impact of VA in HD. It included categories on: symptoms, physical function, activities of daily living, emotional impact, family and social relationships, ability to work or attend school, sleep, and vascular access control. Additional questions exploring the timing of VA decision-making and the barriers patients faced in their HD treatment were added to the original questionnaire. ANED finally approved the questionnaire in terms of languages and comprehensibility for patients.
Data collection
Data collection was performed by a single expert interviewer (SL), in the patient’s mother language (Italian), following the principles of Narrative Medicine [6]. SL had previous interactions with participants. The interviews were audio-recorded, transcribed verbatim, and stored in ANED databases.
Analysis
Interview transcripts were analyzed by SL between March and May 2023. A theoretical framework was developed using principles of grounded theory [7]. Consensus on this framework was reached after review by study team members to ensure that findings reflected the full range and depth of the data.
The results of the patients’ narratives were analyzed using grounded theory methodology supported by the MAXQDA software program (version 2022). Coding was based on Nordyke, et al. 2020 [2]. Further domains not included in the Nordyke, et al. 2020 were described if evident from the data. Results were described based on word frequency in the coding segments (domains and subdomains), and variables such as type of center, age, and HD years were provided as descriptive statistics.
Out of 32 total approached, 30 were interviewed. Reasons for non-participation included lack of interest (n = 2). The average interview was 35 minutes (SD 10.2 minutes).
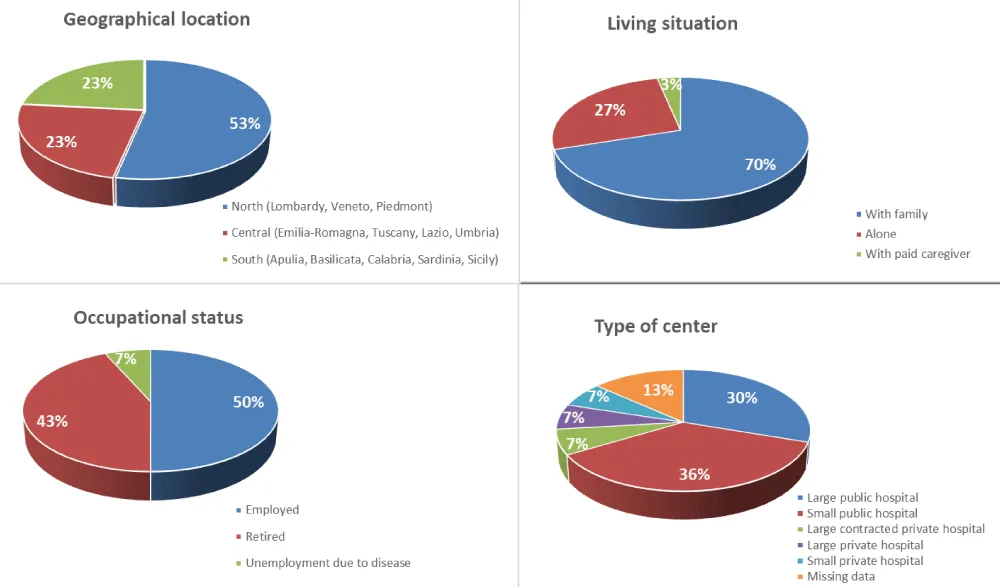
The average age was 61 years (range 20-86 years), and 67% were men. The following characteristics were noted: 27% had previously undergone kidney transplantation; 70% lived with their families; 50% were employed, and 43% were retired. 7% reported being unable to work due to their ongoing dialysis. On average, patients had two vascular accesses before beginning HD (minimum of 1, maximum of 3).
Patients reported that they had been undergoing HD for an average of 9 years (range 1-46) at small public hospitals (37%) or large public hospitals (30%). The remaining cases were managed at large contracted private hospitals (7%), large private hospitals (7%), or small private hospitals (7%). Missing data amounted to 13% (Figure 1).
Figure 1: Sample characteristics.
Vascular access pattern
Variability in the selection of VA is high, and detailed results are provided in Table 1.
| Table 1: Vascular access overview. | |||||
| VA in the past | VA today | N#, % (N=30) | Age, sex | Time on HD (years) | Quotes |
| One VA Type | |||||
| AVF | 9 (30.0%) | 7years, male | 1 | My doctor suggested the fistula as the appropriate option for me, and I agreed. | |
| 65 years, male | 5 | I could not accept anything else, a hose in my neck, never! | |||
| 75 years, male | 6 | My doctor chose the fistula, and I agreed because I preferred it to any other solution. | |||
| 69 years, male | 1 | I do well with the fistula. Nobody told me about other types of access. | |||
| 74 years, male | 5 | My doctor decided… he proposed the fistula… | |||
| 68 years, male | 41 | I have no problem with the fistula. I had a fistula for 41 years. Its duration depended on the center where it was placed. I had it done at a center with expertise. This was important. It allowed me to live normally without any problems. | |||
| 51 years, female | 2.3 | My father's doctor supported me a lot, also from a psychological point of view. At first, they told me about the fistula because of my poor blood circulation. Other options were graft and CVC. | |||
| 55 years, male | 3 | I got a fistula placed before starting the HD. I researched it myself, and I found a better solution for me. | |||
| 47 years, male | 2 | I underwent surgery before starting dialysis. They told me that in my situation, a fistula would be better. I already knew that. | |||
| Two VA types | |||||
| CVC | AVF | 8 (26.7%) | 60 years, male | 5 | Initially, I had a catheter in my neck, then in my chest, and then I got a fistula three years ago, which works perfectly. I underwent invasive surgery for the catheter in my neck… |
| 66 years, male | 19 | The first AVF closed due to thrombi, then the second fistula. As the fistula matured, they inserted the catheter, and when the fistula thrombosed, they placed the catheter back in my jugular. Then I had a transplant after one year, and it closed because my arm was swollen. Then I started dialysis again and got another fistula. | |||
| 54 years, female | 1.2 | I had a CVC with two infections, then I got the fistula. | |||
| 45 years, male | 30 (8 after transplant failure) | The first fistula in my left forearm was 30 years ago. I had the transplant after 3 years; they displaced the fistula. After the rejection, the fistula was placed at the elbow level. Another transplant lasted 13 years, and then they closed the fistula. Then I resumed dialysis, and they placed the fistula in my wrist, which still works. | |||
| 66 years, male | 6 | Emergency CVC until the fistula matures. | |||
| 56 years, female | 6 | From the CVC in the chest to the jugular fistula: it took 3 months to get it. Later, I had the problem of thrombosis and had to have a stent put in. It took 6 months for it to start working properly. | |||
| 59 years, male | 1.5 | I got a central CVC as the fistula matured. | |||
| 63 years, male | 1 | Emergency CVC until the fistula matures. | |||
| AVF | CVC | 4 (13.3%) | 20 years, female | 2 | I had to give up the fistula because it closed. I have a CVC, but I would prefer a fistula because it leaves you more freedom. |
| 86 years, male | 2 | I gave up the fistula due to infection. | |||
| 44 years, female | 5 | The fistula did not mature, so a CVC was inserted as an emergency measure | |||
| 69 years, female | 6 | The fistula in August 2017 did not work; a second one in 2021 worked perfectly. So far, they have not removed the CVC. I still have it. | |||
| AVF | AVF | 1 (3.33%) | 56 years, male | 3 | When I had to start dialysis again, I was angry. Even before the transplant, I had a fistula that did not work; I had to have another one. |
| AVF | AVG | 1 (3.33%) | 64 years, male | 10 | I had a fistula in my right arm. It was not a pleasant experience because I have small veins, and it took several attempts to implant the fistula. After the transplant, I had a graft implanted. The graft was removed after three years and re-implanted. There are no problems with the new graft in my upper arm; it works. |
| AVG | CVC | 1 (3.33%) | 53 years, female | 20 | I couldn't have a fistula because none of my veins were suitable. Then I had a graft in my right forearm, but it closed. Then, in the left forearm, it was still closed. Then I had another one above the elbow, and now a CVC has been in place for 16 years. |
| CVC | AVG | 1 (3.33%) | 83 years, male | 3 | I had a CVC in my leg, which worked for a year, then I had an infection, and then a graft. The doctor wanted to switch to a fistula, but I didn't want a swollen arm. |
| Jugular CVC | Femoral CVC | 1 (3.33%) | 85 years, female, caregiver | NA | The jugular catheter resulted in an infection; now she has a femoral CVC. |
| Three VA types | |||||
| AVF, CVC | AVF | 2 (6.7%) | 72 years, female, caregiver | 15 | The fistula was placed 4 times, and in the meantime, a CVC was inserted because the fistula did not work. She had an inguinal and jugular catheter. The course was turbulent. She had her first fistula in October, and in February, she started dialysis. Then, over the years, it was fine for the first 5 - 6 years until obstructions began to develop. She had to again have a fistula, and, in the meantime, they have inserted a catheter. |
| 62 years, male | 12 | The fistula closed, and so I switched to a temporary catheter. A catheter was inserted, and I got an infection once, and a second time, a thrombus. Then another fistula was implanted. | |||
| CVC, AVG | AVF | 1 (3.33%) | 44 years, male | 2 | I started with an emergency CVC; after 9 months, it became infected, then they placed a graft in March 2022, which didn’t last long because I had too many punctures at the same site. Then, in September 2022, they placed a fistula, but it failed to mature, and I had to take a catheter, which immediately got infected. I had a jugular catheter and, at one time, also a catheter in my groin for 15 days. The graft lasted 6 months, while I expected it to last one year. With the graft, the blood clotted. The catheter is the least invasive as far as the attachment, but it is also the most dangerous because of infections. The catheter is handled by the nurses, and I picked up the infections at the hospital. The graft is a compromise because it does not physically modify the arm. In my case, it didn't last very long. The fistula alters the arm: I can see the arm throbbing. I don't have aneurysms in my arm because I recently got the fistula, but those who have had them have a monstrous arm. I think the best situation is the native fistula. |
| AVF, AVF | CVC | 1 (3.33%) | 57 years, female | 46 | I started dialysis in 1977, starting with peritoneal dialysis. I had three transplants, but the first two went very badly. The last one lasted 9 years. I had the fistula implanted in 1976, and it lasted 27 years. Now I undergo dialysis with a permanent catheter in the jugular. The first fistula lasted 27 years. For 10 years, my father performed the punctures for me, and he never lost a vein. It was removed during the last transplant because of thrombosis, and after that, they inserted a CVC and set up another fistula, which ultimately did not mature. My arm was swollen, I had angina pectoris, and they saved my hand, but I couldn't move it for a while. |
Of the 30 patients interviewed, nine (30.0%) had only one AVF and been on dialysis for an average of 7.8 years (range 1-41 years). In three cases, the fistula was created beforehand due to underlying conditions (such as polycystic kidney) and subsequently mandated HD. One patient reported a positive experience with the fistula for 41 years, attributing its success to the expertise of the center performing the first access and the training provided to patients by hospital staff.
A total of 26.7% started on a CVC and switched to an AVF; they received dialysis on average for 8.7 years (range 1-30 years). In this cohort, the catheter was placed as an emergency measure or temporarily until the fistula matured (for a period ranging between 20 days and 4 months). In addition, the CVC was removed before fistula maturation if an infection or thrombosis occurred. In patients with a long history of dialysis and kidney transplantation, the fistula was closed despite patient preference due to concerns about potential cardiac problems.
A total of 13.3% of patients switched from AVF to CVC (average dialysis time 4 years, range 2-6 years) due to non-maturation and/or infection.
Only 10.0% of patients are currently receiving HD, or previously had a graft. Only one case had been explicitly chosen by the patient. In the other cases, the choice was dictated by clinical circumstances (such as a fistula being inappropriate). Lastly, two patients (6.7%) had a vascular history with two CVC or two AVF as the only access type. The remaining 13.3% reported a more complex history: problems from all types of access, such as CVC infection, graft closure, fistula thrombosis, and potential limb loss. In all the cases, patients complained of failures and inefficiency in the pattern of care. The use of stents was very limited: only one patient aged 56 years with AVF, who visited a large public hospital, and was experiencing renal transplant rejection after three years of treatment, received stenting
Vascular access selection
Patients appear to be aware of the reasons and steps behind the selection of the VA type, but the choice tends to be guided by hospital teams that are following protocols, organizational motives (i.e., CVC is the choice in emergencies), or by assessing the vascular morphology of the patient with scarce consideration of their lifestyle. In most cases, the patients had consciously and confidently accepted the type of access proposed by the center. The patients nevertheless reported that their expectations about access had been met (only 1 out of 30 complained of short-term graft durability). This behavior is likely explained by disease acceptance and the mandatory, lifesaving, permanent nature of HD.
Delays in switching from CVC to AVF/AVG and vice versa were specifically reported by three patients, regardless of the number of years on HD or the type of VA. When asked about their preference, patients reported preferring fistulas, with one patient mentioning grafts and no mention of CVC. AVF was preferred regardless of years on HD or geographical location. Native fistula was mentioned as preferable. A qualitative analysis from patient narratives reported concerns about:
- CVC: potential infections
- AVF: infection, thrombosis, bumping, swelling, pressure, and cannulation (needle size)
- AVG: positioning, duration, and maturation
Patient populations were variable in relation to sex, age, and factors such as year on HD, type of HD, and location of HD center.
A qualitative analysis from patient narratives reported that patients mostly collect information from physicians and the web; their patient support groups also play a relevant role.
| Illustrative quotes |
| AVF |
| I can wash and have an almost normal life - 64 years,male |
| The surgery was difficult, I felt no pain, but I felt the fistula functioning. My arm didn't swell, and you can hardly see that I'm on dialysis. You have to puncture every day - some nurses are very skilled, and you don’t feel the needle - but then you get used to it - 51 years, female |
| I had a fistula for 41 years. Its duration depends on the clinic performing the fistula. It allowed me to live normally without any problems - 68 years, male |
| CVC |
| CVC is more bothersome than a fistula because of big patches, I experienced itchy skin - 63 years, male |
| The CVC is outside the body; therefore, it is more bothersome, it prevents some daily activities - 74 years, male |
| A jugular vein CVC was temporarily placed to give the fistula time to mature - 66 years, male |
| With CVC you have the problem of dressings; besides, protocols vary a lot - 44 years, female |
| With CVC you don't have to repeatedly puncture as with the fistula. I need to be careful to avoid infections - 20 years, female |
| AVG |
| The implant is invisible and causes no problems. I got it after the transplant. The implant was removed and replaced after three years. The new implant in the upper arm causes no problems, and now my access is good - 64 years, male |
| Then I had an implant in my right forearm, but it closed. Then on the left, and it still closed. Then I had another one above the elbow, and now I have a CVC - 53 years, female |
| The implant definitely makes me more independent; I feel better - 83 years, male |
Patient populations were variable in relation to sex, age, and factors such as year on HD, type of HD, and location of HD center. Analysis from patient narratives reported that the patient experienced VA-related adverse events (Table 2). Skin disorders are reported to be mostly related to skin sensitivity. Cases of thrombosis, potentially causing steal syndrome in the most severe instances, were mostly resolved by removing the AVF and replacing it with a CVC; stenting was only used in one case. Episodes of infections were reported when CVC was used, prompting physicians to switch to AVF. CVC was replaced with a graft in only one instance. Physicians switched from AVF to CVC when infections or thrombosis occurred, apparently regardless of the patient’s clinical history or preference. This is confirmed in two interviews with patients with complex clinical histories, both having undergone transplantation and having a long history of HD. In one case, stenting solved the adverse event and enabled HD at home (with patient satisfaction); in the other case, the AVF failed, and a CVC was ultimately used (with patient discontent).
| Table 2: Vascular access adverse events by center type and region. | |
| Center type | Adverse events |
| Large public hospital | Infections |
| Skin disorders | |
| Thrombosis | |
| Small public hospital | Infections |
| Skin disorders | |
| Thrombosis | |
| Large private contracted hospital | Infections |
| Large private hospital | Infections |
Experience at hemodialysis centers
Patients expressed concerns about various aspects of HD centers, regardless of center type or duration of HD treatment. Issues included difficulties in accessing centers due to parking shortages, lack of dedicated spaces, and the inability to undergo HD at home. Some patients felt inadequately supported by physicians (lack of empathy) and/or nurses (lack of focus on patient training for daily life with venous access). Others noted a lack of psychological support and dissatisfaction with inadequate CVC dressing protocols that were not focused on the patients’ needs. Conversely, positive experiences were often reported with small public hospitals where patients received appropriate and empathetic support from physicians and nurses (e.g., training with blunt needles) (Table 3).
| Table 3: Patients' experience at the hemodialysis center. | |||
| Quotes | Sex | Years of HD, current VA type | Region/center type |
| Negative center aspects | |||
| AVF | |||
| I would like to emphasize the experience of the psychologist, in collaboration with ANED, which was important for me. During the pandemic, however, it unfortunately stopped. | male | 5 | Emilia-Romagna Large public hospital |
| The waiting time to place the fistula was exceptionally long. The problem stemmed from the lack of a vascular surgeon dedicated to fistulas at the center. The pre-dialysis aspect is important when you start doing the protein diet, which is not reimbursed by the local health unit |
female | 15 | Lombardy Large contracted hospital |
| I hope there will be more attention to those who go to the hospital and the dedicated parking lots. | male | 12 | Veneto Large public hospital |
| I would like to emphasize the importance of home hemodialysis. It is not widely utilized, yet I find it beneficial both in avoiding hospital transfers and providing flexibility. After dialysis, I feel well and can carry out daily activities without any issues, while during dialysis, my blood pressure drops. Traveling has a significant impact on me. | female | 6 | Lombardy Large public hospital |
| I did not receive any information, and there is a lack of concern for patient education. | female | 1.2 | Sicily Small public hospital |
| The logistical aspect, traveling and getting around, is difficult; you must organize yourself. | male | 6 | Piedmont Large public hospital |
| In the past, health care professionals provided instructions, explanations, and information. I was trained on everything: the dialysis process and what to do in case of emergency and nurse absence, puncturing myself, and fistula management. I participated in my pattern of care. Now this doesn't happen anymore. | male | 41 | Piedmont Large public hospital |
| With a fistula, dialysis is better, even though the chelators they use are obsolete. | male | 2 | Lombardy Small public hospital |
| Dialysis is essential to my life, but I did not receive adequate support from physicians, as they lacked empathy. | male | 5 | Sardinia Small public hospital |
| I trust my nephrologist very much. Doctors are often not very focused on the human aspect. | male | 3 | Emilia-Romagna Large public hospital |
| CVC | |||
| The logistics must be planned, and there is too much bureaucracy to move to another center. | female | 6 | Lombardy Small private hospital |
| With the catheter, you have the problem of dressings, because each hospital has its own protocol: in some hospitals, they used to use smaller, more manageable dressings. Now they are bigger and more difficult to manage. | female | 5 | Lombardy Large public hospital |
| Positive center aspects | |||
| AVF | |||
| At my current center, I am experiencing positive outcomes. The blunt needle is preferable to the traditional needle because it minimizes discomfort and does not cause tearing. | male | 5 | Emilia-Romagna Large public hospital |
| I feel particularly good at this hospital, like part of the family. | male | 1 | Umbria Small public hospital |
| The fistula was implanted some time before dialysis, and I prepared myself mentally and physically. | female | 2.2 | Lombardy Small public hospital |
| I had a fistula for 41 years. The duration depends on the clinic implanting the fistula. Having an expert center allowed me to live normally without any problems. | male | 41 | Lombardy Small public hospital |
| AVG | |||
| At my center, both doctors and nurses take care of me. We are also supported by psychologists. | male | 10 | Emilia-Romagna Small public hospital |
Vascular access-specific health-related quality of life impact
Health-Related Quality of Life (HRQoL) was assessed according to Nordyke, et al. 2020 [4]. Six domains and 21 subdomains were initially defined, but an additional 11 subdomains emerged from the research interviews. These additional subdomains were: namely attention, carrying weights, change in diet and cooking, caregiver, rage, embarrassment, resignation/acceptance, dizziness/pressure disorders, limitation of Activities of Daily Living (ADLs), flushing/itching, and impact with others.
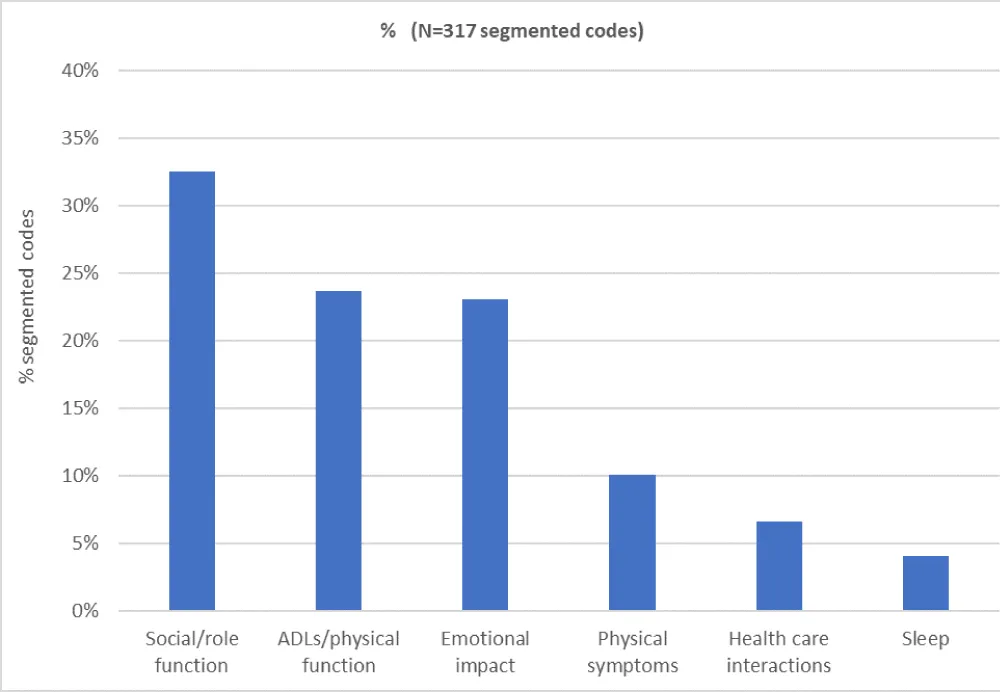
A total of 317 segmented codes were analyzed to assess the impact of VA on patient HRQoL. Results are depicted in Figure 2.
Figure 2: Negative impact of vascular access on patient HRQoL.
The social/role function (32% of segmented codes in all documents) was mainly influenced by recreation and travel (21%), family impact/play (19%), and work/school (18%). Time burden (15%), relationships with others (9%), appearance/clothing (8%), social avoidance (7%), and physical intimacy (3%) were also notable.
The burden of ADLs/physical function (24%) included housework (25%), attention to daily activities (23%), showers/hygiene, carrying weight, and change in diet and cooking (each 15%), and caregiver (8%).
The emotional impact (23%) encompassed resignation/acceptance (40%), worry/anxiety (23%), fear (22%), embarrassment (11%), and rage (4%).
Vascular accesses had less impact on physical symptoms (10%), with patients reporting manageable bruising/swelling, dizziness/pressure disorders, and pain on cannulation or limitation in movement. Health care interactions (7%) and sleep (4%) were of lesser impact.
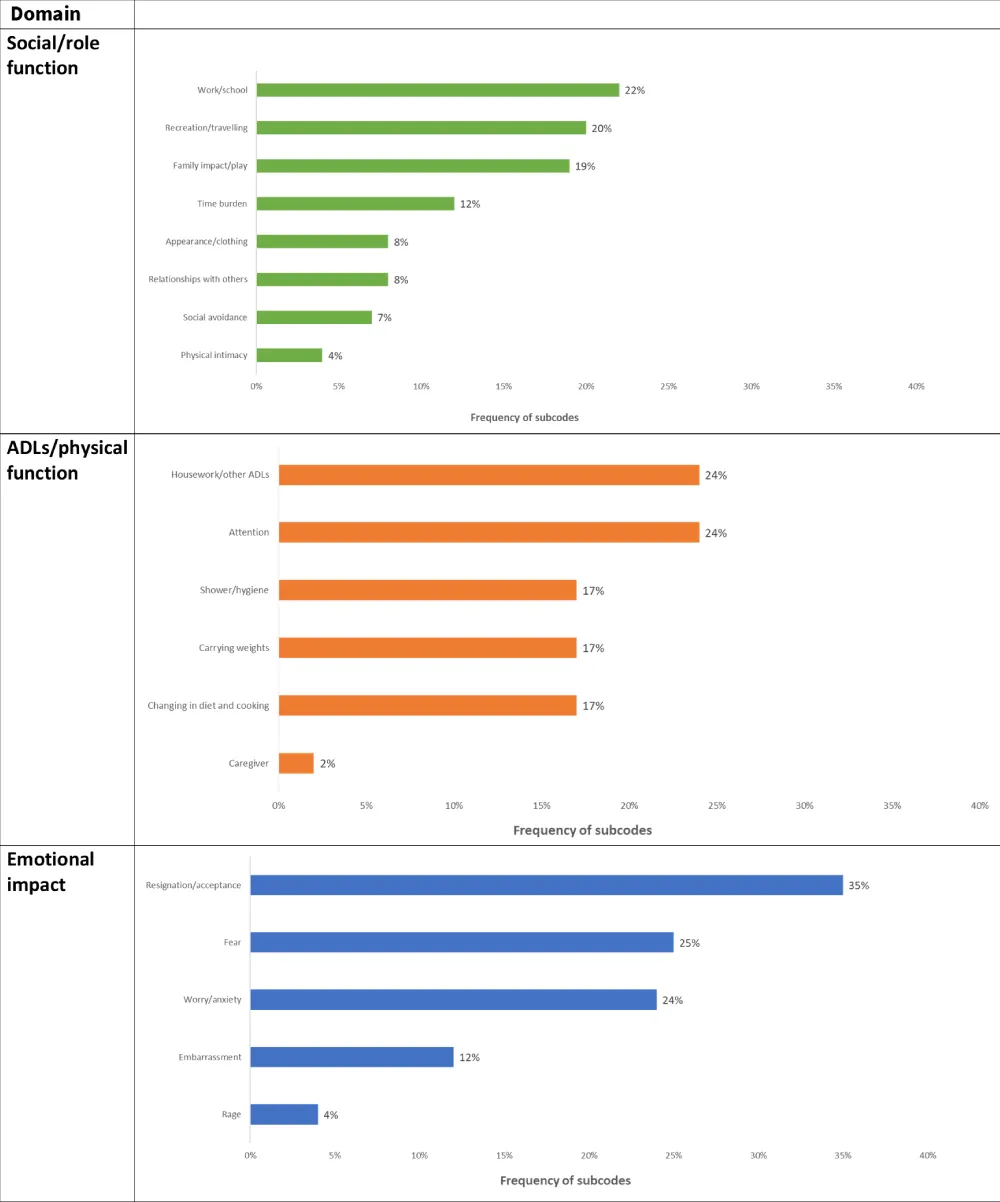
Younger patients (≤65 years, i.e., 60% of participants) experienced a major impact on work, recreation/travel, and family subdomains. They suffered particularly from impairments relating to homework/ADLs and attention subdomains, as well as feelings of fear, anxiety/worry, but mostly resignation/acceptance (Figure 3).
Figure 3: Frequency of subcodes in the first three domains and related subdomains (Social/role function, ADLs/physical function, Emotional impact) among patients ≤65 years.
Regarding the three most relevant domains, the results are comparable with those reported by Nordyke et al. 2020. However, differences in the social/role function, ADLs/physical function, and emotional impact were observed, particularly in appearance/clothing (8 mentions versus 33 mentions, respectively), social avoidance (7 versus 29, respectively), and work/play (19 versus 5, respectively). These variations in worry/anxiety (17 versus 75, respectively) may likely be attributed to differences in the sample characteristics.
Which type of access had a greater impact on the most relevant domains?
From patient narratives, AVF is reported to negatively affect patients’ lives more than other types of vascular access.
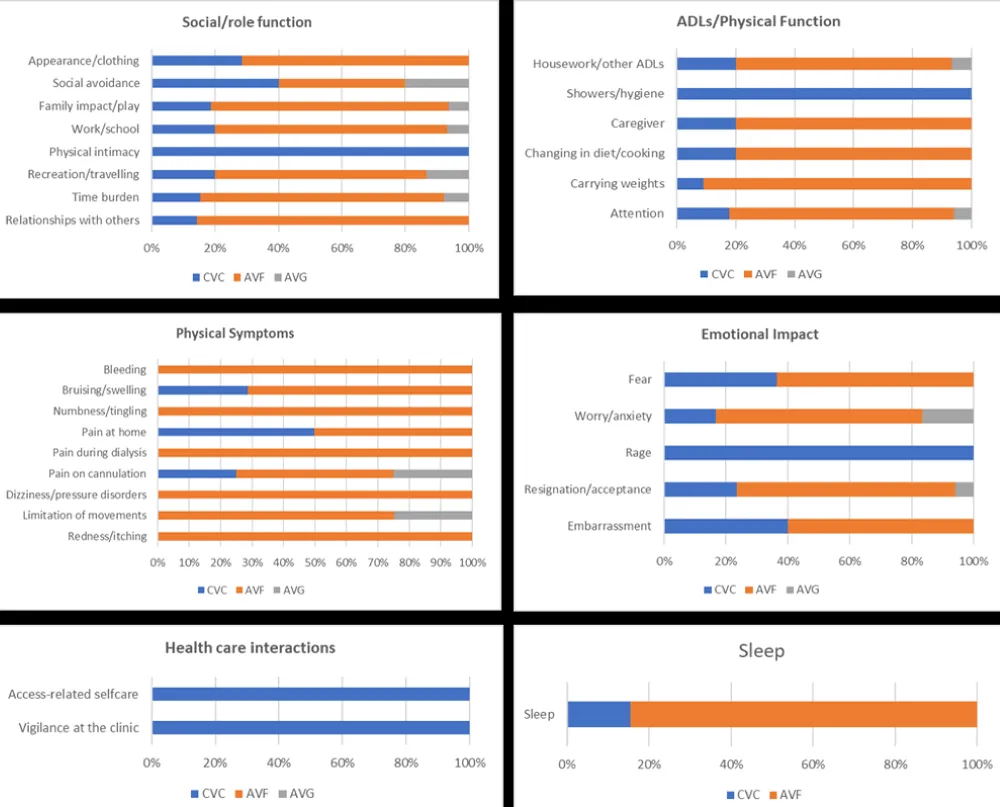
As reported in Figure 4, AVF had a greater adverse impact than CVC and AVG on the social/role function, particularly in terms of relationships with others, time burden, recreation/travel, work/school, and family impact/play. Additionally, AVF had a greater adverse impact than CVC on appearance/clothing, as patients had to conceal their AVF more frequently due to visible swelling. The impact of AVG on the social/role function domain was extremely low.
AVF also affected ADLs/physical function more than CVC and AVG in terms of attention, carrying weights, and caregiver requirements. Patients with AVF who required caregiver assistance were often older (>80 years) or, if younger (>50 years), preferred support with ADLs. However, experiences involving diet and cooking were reported in relation to HD rather than vascular access. An impact on showers/hygiene was reported only for CVC. Housework and other ADLs were mostly reported to be more difficult due to HD rather than vascular access; however, AVF posed a risk of contamination (e.g., when cooking), and CVC posed a risk if accessories were dropped.
Furthermore, AVF had a greater impact on the emotional domain compared to CVC and AVG. Feelings of resignation/acceptance were often associated with HD and, consequently, the impact of vascular access. Embarrassment affected more patients with AVF due to curiosity from others concerning AVF swelling. Worry/anxiety and fear were also greater and primarily linked to the duration of treatment and infections or AVF failure. AVF also affected sleep due to limited arm motion, while patients with CVC needed to consider the impact of access-related self-care due to the size of bandages and patches.
Figure 4 illustrates the impact of vascular access on health domains.
Figure 4: Impact of vascular access on patients' lives – analysis by domain and subdomains (weighted segments values).
Regarding physical symptoms, such as bleeding, numbness/tingling, pain during dialysis, dizziness/pressure disorders, and redness/itching, the impact of AVF was greater than CVC and AVG. The lowest rate of physical symptoms was reported for AVG.
| Illustrative quotes: |
| SOCIAL/ROLE FUNCTION |
| Relationships with others |
| VA has a significant impact on daily life and relationships, especially with strangers - 20 years, female, CVC user |
| People often ask, and it bothers me; they inquire, "what do you have there?" - 65 years, male, AVF user |
| My friends are aware; by now, they are accustomed to seeing my swollen arm - 75 years, male, AVF user |
| My whole life changed, and the VA is just a part of that - 59 years, male, AVF user |
| Recreation/travel |
| I couldn't play volleyball - 20 years, female, CVC user |
| I couldn't move; it's always tied to dialysis and then figuring out where to do it: you need to see if there is a center…72 years, female, AVF user |
| I can't have a relaxed vacation; I always need a referral hospital for dialysis - 60 years, male, AVF user |
| Work/school |
| Working alone is really problematic, and I'm about to close down - 62 years, male, AVF user |
| I must take time off work, do dialysis and then restart. I have a very understanding director. It’s a restriction to my daily life during which I am absent, but you need to be organized - 56 years, female, AVF user |
| When I was working, it wasn't easy, and I was stalked by a boss. People don't understand - 57 years, female, CVC user |
| Family impact/play |
| It impacted my life and my husband's - 72 years, female, AVF user |
| My wife had to stop working to support me. It is not easy, either for me or for her - 59 years, male, AVF user |
| Appearance/clothing |
| In winter, I have no problem because I can hide it well; in summer I can’t - 20 years, female, CVC user |
| I have to cover the fistula because people don't understand; I can't stand having to explain - 45 years, male, AVF user |
| When I was young, I had no problem wearing a short-sleeved shirt; later, I couldn’t - 57 years, female, CVC user |
| Physical intimacy |
| The catheter doesn't help with my self-esteem. It's a bulky presence. As a woman, it's hard to like yourself - 57 years, female, CVC user |
| ADLS/PHYSICAL FUNCTION |
| Attention |
| I try to do what I used to do but with more attention and no effort - 71 years, male, AVF user |
| Carrying weights |
| I cannot carry weights - 72 years, female, AVF user |
| Eating |
| Some foods should be avoided and you need a dietitian. I found leaflets from ANED with tables of the foods that can be eaten - female, 44 years, CVC user |
| Examples of quotes: |
| Showers/hygiene |
| I try to protect it when I shower. Now I have a new shower patch- female, 53 years, CVC user |
| When I shave, I have to be careful - 86 years, male, CVC user |
| With the CVC everything was complicated, even washing - male, 45 years, AVF user |
| With the CVC it is more difficult washing, I need help from a family member - male, 59 years, AVF user |
| EMOTIONAL FUNCTION |
| Embarrassment |
| The first time, when the fistula was placed, I thought that it was the last time I would see my arm like that - male, 68 years, AVF user |
| I must conceal the fistula because people don't understand, I can't stand having to explain. The visual impact is very strong - male, 45 years, AVF user |
| I try to avoid because people stare at me. You feel discomfort - female, 57 years, CVC user |
| Worry/anxiety |
| The duration is very important, it also affects your mood - male, 65 years, AVF user |
| Infections are worrying but I don't think about it - female, 54 years, AVF user |
| Duration is not easy to consider, it makes me anxious - male, 44 years, AVF user |
| CVC infections worry me because they made me sick - female, 57 years, AVF user |
| I had problems with a fistula because it closed and couldn't mature - male, 64 years, AVG user |
| I was informed about the duration issue: You must change the implant position after a while. Placement of the implant is not easy for me because of my small veins (3-4 Dopplers were performed) - male, 83 years, AVG user |
| Fear |
| I can take care of my VA, but I am terrified about potential emergencies. It's a precarious and uncertain situation that I try not to think about. It's like the sword of Damocles. I feel terrified and afraid - male 65 years, AVF user |
| Anguish and fear are my main feelings - female, 72 years, AVF user |
| Fear is also relative to the duration of the fistula; I don't want to watch while they stick the needle in. I suffered from panic attacks but fortunately they have stopped - male, 44 years, AVF user |
As reported in international data [4], vascular access impacts HRQoL in Italian HD patients with ESKD, mostly in the social, ADLs/physical function, and emotional domains.
Unlike other cultural contexts, acceptance of and resignation to lifelong HD and the increased attention needed in activities of daily living with VA were added as subdomains of impairment by Italian patients. Common subdomains of major concern to patients are time burden, recreation/travel, and fear and worry/anxiety concerning infections and the duration of use of medical devices. In terms of patterns, patients mostly accept the decisions made by health care providers on VA selection, relying on and trusting in their choice. Their preferences do not appear to be considered, with all participants reporting a preference for AVF and none preferring CVC. Though at present, CVC is still largely used in clinical practice (10% reported a delay in CVC removal). Besides, the 2019-K-DOQI guidelines [2] indicate that the patient’s clinical evaluation (i.e., microcirculation) should be performed, yet not always experienced in the CVC implant, as reported from patients’ words. The preference for home HD is never discussed, nor is the possibility of stenting in case of thrombosis. Consequently, VA history could become more complex, and VA patterns prove to be beyond the patient’s control. As an example, the fistula could be locked after a renal transplant and needs to be reopened in case of transplant rejection, thus significantly impacting quality of life. Furthermore, appropriate care concepts impact the quality of the VA experience: for example, a native fistula and patient training on fistula management could boost the probability of a longer fistula duration and increase patient reassurance. The results are consistent with recent literature findings: HRQoL has been shown to predict mortality in patients receiving maintenance HD; VA is critical in these patients. Multivariable analyses showed that the overall score, social functioning score, and the dialysis-related complication score of the VA questionnaire influenced HRQoL in the study population [6]. Furthermore, total HRQoL scores and scores on the physical and mental component domains were significantly higher in the satisfied VA group than in the dissatisfied group at baseline. Wu, et al. 2024 data also suggested a significant association between VA satisfaction and HRQoL in maintenance HD patients. Surgeons and nephrologists should therefore incorporate patient satisfaction into VA surgical decision-making [8].
This research did not specifically include patients from surgical hubs; the research objective reflected the real-life variability of center types. This could imply increasing heterogeneity in VA protocols. Nevertheless, the pattern of VA use in this study partially matches results obtained from other research on 3,451 HD patients in regional administrative databases representative of the Italian territory [4]: CVC use prevailed (33.1% CVC, 14.7% AVF, and 0.8% AVG) when a single VA was used (30% used AVF in this research); in case of more than one VA type, 20% - 25% of patients alternate CVC/AVF use in both studies, while AVG is mostly selected after experiencing CVC/AVF. These patterns suggest partial application of the 2019-K-DOQI guidelines recommending arteriovenous access (AVF or AVG) in preference to CVC in most incidents and prevalent HD cases due to the lower infection risk associated with VA use [2]. Long-term CVC use is recommended only if multiple prior arteriovenous accesses failed with no available options or valid patient preference, and when the use of an arteriovenous access would severely limit QoL or achievement of life goals. Patients should be properly informed of patient-specific risks and the benefits of potential and reasonable access options. In general, no decision about a single VA creation or placement should be made in isolation or independent of the patient’s overall ESKD life plan. However, this study demonstrated a misalignment between clinical practice and patient preferences and experience, as demonstrated by the large use of AVF, which is the VA with a major impact on QoL, the delay in the CVC removal, and the lack of acknowledgment about the graft.
Qualitative research was selected as the preferred method for describing the impact of health care technologies and/or treatments on patients’ lives, based on the assumption that narration is an essential human prerequisite and is part of the health care pattern [9]. This could be considered a limitation in the representativeness of the study sample for ESKD patients. However, the sample size of this research confers with current qualitative literature [4,10]. Robustness is achieved, moreover, through the use of a validated questionnaire [4].
Lastly, recent qualitative literature findings demonstrated that dialysis-related decisions need to involve shared decision-making on VA, and that a timely, tailored, and balanced exchange of information between patients and their care team is required [10]. Research demonstrated that opportunities to revisit the VA decision before and after starting dialysis helped prepare patients for their access and promoted ongoing alignment between patients’ care priorities and treatment plans. Where shared decision-making was undermined, HD via catheter ensued as the most readily available VA option [10].
Active participation of patients, in accordance with their preferences and requirements, should be encouraged due to the significant impact of vascular access on various aspects of a patient’s QoL, encompassing social/role dynamics, activities of daily living, and both physical and emotional functions. This finding underscores the necessity to redefine and thoroughly discuss VA protocols within the scientific community, involving patients in the co-creation process.
Additionally, increasing awareness and use of grafts among patients and healthcare professionals is crucial, as grafts have the potential to significantly improve quality of life. The use of AVG and stenting techniques, compared to AVF and CVC, has been linked to improved quality of life for patients, due to a reduction in infections and complications, as well as better mobility [11-13]. The spread of home HD as a potential treatment option is also to be considered for minimizing the adverse effects on QoL.
Furthermore, enhancing collaboration with patient associations is essential to increasing patient health literacy and awareness regarding VA, thereby facilitating informed decision-making when selecting or changing the VA. Nephrologists should assume a predominant role in the provision of VA care and maintain close interdisciplinary collaboration with other specialties to ensure the delivery of optimal, multidisciplinary patient care [14].
Authors’ contributions
Research idea and Study design: SM, SL; data acquisition: SL; data analysis/interpretation: SL, SM; statistical analysis: SL; supervision or mentorship: SM. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions about the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Support
W. L. GoRe & Associati S.r.l provided financial support for this project.
The funders did not have any role in study design, collection, analysis, interpretation of data, or the decision to submit the report for publication.
Financial disclosure: Dr. Mangano reports serving as a consultant for W. L. Gore & Associati S.r.l and Fresenius. Dr. Lopatriello reports having served as a scientific consultant for several companies, including Helaglobe S.r.l employee at the time of the manuscript drafting.
The need to understand patients’ perspectives on vascular access and its impact on daily life was identified and initiated by W. L. Gore & Associati S.r.l., represented by Dr. Geraldo Tadinho Monteverde Spencer, Dr. Marzia Martoni, and Dr. Francesca Di Stasi, who provided the initial impetus for this project. ANED (Associazione Nazionale Emodializzati Dialisi e Trapianto) played a crucial role in contacting its members and obtaining their informed consent for participation. Helaglobe S.r.l. is acknowledged as the employer of SL, the author of this manuscript.
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. 2024;105(4 Suppl):S117-S314. Available from: https://doi.org/10.1016/j.kint.2023.10.018
- Lok CE, Huber TS, Lee T, Shenoy S, Yevzlin AS, Abreo K, et al. KDOQI clinical practice guideline for vascular access: 2019 update. Am J Kidney Dis. 2020;75(4 Suppl 2):S1-S164. Available from: https://doi.org/10.1053/j.ajkd.2019.12.001
- Woo K, Lok CE. New insights into dialysis vascular access: what is the optimal vascular access type and timing of access creation in CKD and dialysis patients? Clin J Am Soc Nephrol. 2016;11(8):1487-1494. Available from: https://doi.org/10.2215/cjn.02190216
- Nordyke RJ, Nicholson G, Gage SM, Lithgow T, Himmelfarb J, Rivara MB, et al. Vascular access-specific health-related quality of life impacts among hemodialysis patients: qualitative development of the hemodialysis access-related quality of life (HARQ) instrument. BMC Nephrol. 2020;21(1):16. Available from: https://doi.org/10.1186/s12882-020-1683-5
- Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349-357. Available from: https://doi.org/10.1093/intqhc/mzm042
- Charon R. Narrative medicine: a model for empathy, reflection, profession, and trust. JAMA. 2001;286(15):1897-1902. Available from: https://doi.org/10.1001/jama.286.15.1897
- Corbin J, Strauss A. Grounded theory research: procedures, canon, and evaluative criteria. Qual Sociol. 1990;13:3-21. Available from: https://eli.johogo.com/Class/Strauss.pdf
- Wu M, Li MT, Zhang LP, Wei D, Han YC, Gao M. The impact of vascular access satisfaction on health-related quality of life in patients receiving maintenance hemodialysis: a 2-year follow-up study. J Vasc Access. 2024;25(5):1467-1473. Available from: https://doi.org/10.1177/11297298231163224
- Palla I, Turchetti G, Polvani S. Narrative medicine: theory, clinical practice and education - a scoping review. BMC Health Serv Res. 2024;24(1):1116. Available from: https://doi.org/10.1186/s12913-024-11530-x
- Elliott MJ, Ravani P, Quinn RR, Oliver MJ, Love S, MacRae J, et al. Patient and clinician perspectives on shared decision making in vascular access selection: a qualitative study. Am J Kidney Dis. 2023;81(1):48-58.e1. Available from: https://doi.org/10.1053/j.ajkd.2022.05.016
- Aitken E, Thomson P, Bainbridge L, Kasthuri R, Mohr B, Kingsmore D. A randomized controlled trial and cost-effectiveness analysis of early cannulation arteriovenous grafts versus tunneled central venous catheters in patients requiring urgent vascular access for hemodialysis. J Vasc Surg. 2017;65(3):766-774. Available from: https://doi.org/10.1016/j.jvs.2016.10.103
- Woo K, Ulloa J, Allon M, Carsten CG 3rd, Chemla ES, Henry ML, et al. Establishing patient-specific criteria for selecting the optimal upper extremity vascular access procedure. J Vasc Surg. 2017;65(4):1089-1103.e1. Available from: https://doi.org/10.1016/j.jvs.2016.10.099
- Kingsmore DB, Stevenson KS, Thomson PC, Kasthuri R, Knight S, Jackson A, et al. Pre-emptive or reactive treatment, angioplasty or stent-graft? The outcome for interventions for venous stenosis in early-cannulation arteriovenous grafts. J Vasc Access. 2023;24(2):253-260. Available from: https://doi.org/10.1177/11297298211029413
- Hassanein M, Radhakrishnan Y, Hernandez Garcilazo N, Khor SY, Norouzi S, Anvari E, et al. Global hemodialysis vascular access care: three decades of evolution. J Vasc Access. 2025;26(1):5-14. Available from: https://doi.org/10.1177/11297298231159251