More Information
Submitted: March 04, 2026 | Accepted: March 12, 2026 | Published: March 13, 2026
Citation: Boumaiz F, Zniber A, Sakout N, Kadiri N, Ouzeddoun N, Benamar L. Predictors of Complications and CKD Progression in Adult Nephrotic Syndrome: A Cohort Study. J Clini Nephrol. 2026; 10(3): 032-038. Available from:
https://dx.doi.org/10.29328/journal.jcn.1001173
DOI: 10.29328/journal.jcn.1001173
Copyright license: © 2026 Boumaiz F, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Nephrotic syndrome; Glomerular disease; Chronic kidney disease; Complications
Predictors of Complications and CKD Progression in Adult Nephrotic Syndrome: A Cohort Study
Firdaous Boumaiz* , Amal Zniber, Nadia Sakout, Nada Kadiri, Naima Ouzeddoun and Loubna Benamar
, Amal Zniber, Nadia Sakout, Nada Kadiri, Naima Ouzeddoun and Loubna Benamar
Nephrology Department, Ibn Sina University Hospital, Rabat, Morocco
*Corresponding author: Firdaous Boumaiz, Nephrology Department, Ibn Sina University Hospital, Rabat, Morocco, Email: [email protected]
Background and aims: Nephrotic syndrome (NS) is a glomerular syndrome characterized by massive proteinuria and severe complications, including chronic kidney disease (CKD). This study aimed to describe the demographic, clinical, biochemical, histological, and etiological characteristics of patients with NS, to identify risk factors for complications, and to assess predictors of progression to CKD in a Moroccan setting.
Methods: This retrospective, descriptive, and analytical study was conducted in the Nephrology Department of CHU Ibn Sina, Rabat, Morocco, from 2019 to 2024. It included 308 patients hospitalized for NS, aged >15 years, with proteinuria ≥3.5 g/day and hypoalbuminemia < 30 g/L. Data were extracted from medical records and analyzed using Jamovi software. Univariate and multivariable analyses (logistic regression) were performed; p < 0.05 was considered statistically significant.
Results: Mean age was 42.5 ± 15 years, with a sex ratio of 1.03. An edematous syndrome was present in 65.76% of patients, and hypertension in 35%. Median proteinuria was 7 g/24 h, and median serum albumin was 20 g/L. Histologically, focal segmental glomerulosclerosis (FSGS) was the most frequent lesion (26%), followed by lupus nephritis (21%). Primary NS accounted for 50.7% of cases. Complications occurred in 43.4% of patients, including acute kidney injury (36%), infections (10%), and thromboembolic events (3.5%). Complete remission was observed in 54% of patients, while 21% progressed to CKD. Admission serum creatinine was significantly associated with progression to CKD (p = 0.001).
Conclusion: Nephrotic syndrome represents a major clinical burden in nephrology, with a predominance of FSGS and a high rate of CKD. Early detection, multidisciplinary management, and prophylactic interventions are essential to reduce morbidity and slow renal progression. These findings highlight the need for further research and tailored health policies.
Nephrotic syndrome (NS) is a glomerular syndrome characterized by a set of biological abnormalities leading to significant plasma disturbances [1]. According to KDIGO, it is biologically defined by the association of three main criteria: persistent, massive proteinuria (>3.5 g/day) and hypoalbuminemia (< 30 g/L), and/or hypoproteinemia (< 60 g/L) [2].
In adults, the causes of nephrotic syndrome are diverse, ranging from primary glomerular diseases, such as minimal change disease and focal segmental glomerulosclerosis, to secondary glomerular diseases (diabetes, systemic lupus erythematosus, infections, etc.) [3].
Complications are frequent and potentially severe, including an increased risk of infections, thromboembolic complications, and metabolic disorders such as dyslipidemia [1].
The clinical course of NS is variable, ranging from spontaneous or treatment-induced remission to progression to chronic kidney disease (CKD) and even end-stage kidney disease (ESKD) [1].
Given the high frequency, severity, and etiological heterogeneity of nephrotic syndrome, as well as the absence of a dedicated national registry, an in-depth evaluation of this syndrome is warranted [4-6].
In this study, we aimed to:
- Describe the demographic, clinical, biochemical, histological, and etiological characteristics of patients with nephrotic syndrome.
- Determine risk factors for developing nephrotic syndrome complications.
- Evaluate risk factors for progression to chronic kidney disease.
Study design
This was a retrospective, descriptive, and analytical study conducted in the Nephrology Department of CHU Ibn Sina, Rabat, Morocco, over the period 2019–2024. This design allowed assessment of the clinical profiles and outcomes of nephrotic syndrome in this patient population. The descriptive component aimed to highlight the epidemiological, clinical, and biochemical characteristics of affected patients, whereas the analytical component explored factors associated with disease progression. In addition, the single-center design ensured good homogeneity of diagnostic and therapeutic practices.
Study population
All patients hospitalized for nephrotic syndrome in the Nephrology Department of CHU Ibn Sina, Rabat, between 2019 and 2024, aged >15 years, were considered, according to the following inclusion and exclusion criteria.
Inclusion criteria:
- Age >15 years.
- Confirmed diagnosis of nephrotic syndrome, based on:
- 24-hour proteinuria ≥3.5 g/day or a protein-to-creatinine ratio ≥3 g/g, and
- Serum albumin <30 g/L.
- Follow-up duration ≥1 year.
Exclusion criteria:
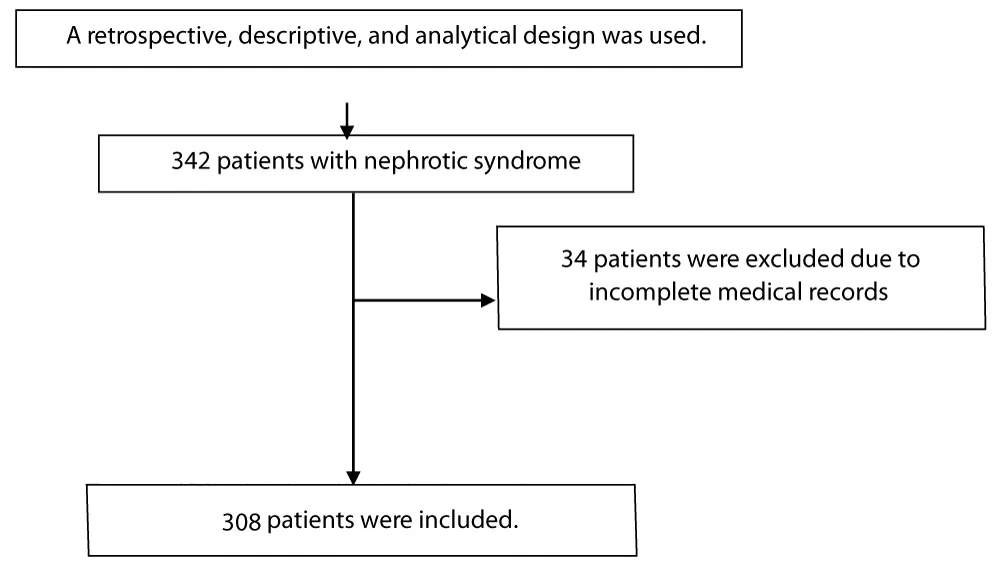
- Incomplete medical records (34 patients were excluded for this reason).
- Ongoing infection, which could interfere with the interpretation of laboratory findings (Figure 1).
Figure 1: Flow diagram summarizing the study methodology.
Study design and data collection
We conducted a retrospective study including patients diagnosed with nephrotic syndrome between January 2019 and December 2024. Data were extracted from medical records and hospital databases.
Variables
- Demographics: age, sex, city of residence, health insurance/coverage status.
- Clinical: lower-limb edema, hypertension, and urine dipstick findings.
- Laboratory: 24-hour proteinuria, total serum protein, serum albumin, serum creatinine, and estimated glomerular filtration rate (eGFR, mL/min/1.73 m²) using the MDRD equation, urine culture (ECBU), and complete blood count.
- Histology: kidney biopsy (PBR) and minor salivary gland biopsy (BGSA), when performed [7].
Statistical analysis
Analyses were performed using Jamovi.
Categorical variables were reported as n (%). Continuous variables were reported as mean ± SD when normally distributed or median (IQR) otherwise. Univariate comparisons used parametric or non-parametric tests as appropriate. Factors associated with progression to chronic kidney disease (CKD) were assessed using univariate and multivariable logistic regression. Statistical significance was set at p < 0.05.
Sex
A total of 308 patients with nephrotic syndrome were included. The male-to-female ratio was 1.03.
Age
Mean age was 42.5 ± 15 years (range 15–85). Age distribution: 25–64 years (72%), 15–24 years (18%), 65–74 years (8%), and >75 years (2%).
Medical history and comorbidities
The most frequently reported medical history and nephrology-relevant comorbidities were hypertension, diabetes mellitus, and substance use (tobacco, alcohol, and cannabis). Overall, 43% of patients had at least one prior medical history, while 57% reported none (Table 1).
| Table 1: Main comorbidities and past medical history (N = 308). | |
| Past medical history | n (%) |
| Hypertension | 71 (23.0) |
| Substance use (tobacco/alcohol/cannabis) | 37 (12.0) |
| Diabetes mellitus | 26 (8.4) |
| Systemic lupus erythematosus (SLE) | 18 (5.8) |
| Heart disease | 12 (3.9) |
| Tuberculosis | 12 (3.9) |
| History of kidney stones | 4 (1.3) |
| Malignancy | 3 (1.0) |
| Abbreviation: SLE, systemic lupus erythematosus. | |
Clinical presentation at admission
At admission, edema was present in 65.76% of patients; anasarca occurred in 5.43%, consistent with advanced nephrotic syndrome and severe hypoalbuminemia (Table 2). All patients had proteinuria on urine dipstick.
| Table 2: Clinical features at admission (N = 308). | ||
| Feature | n | % |
| Lower-limb edema | 202 | 65.76 |
| Anasarca | 15 | 5.43 |
| Proteinuria on dipstick | 308 | 100 |
| Hypertension | 109 | 35 |
Laboratory findings and nephrotic syndrome phenotype
Median 24-hour proteinuria was 7 g/24 h (range 3.5–24). Median serum albumin was 20 g/L (range 8–29), and median total serum protein was 49 g/L (IQR 40–58). Median serum creatinine was 15 mg/L (IQR 3–330).
A nephrotic syndrome with nephritic/complicated features (“impure” nephrotic syndrome) was predominant (256 patients; 83%), with hypertension (35%), renal impairment (36%), and hematuria (67%).
Kidney biopsy and histological diagnoses
Kidney biopsy was performed in 300/308 patients (97.5%). The most frequent diagnosis was focal segmental glomerulosclerosis (FSGS) in 78 patients (26%) (Table 3).
| Table 3: Kidney biopsy findings (N = 300). | |
| Histological diagnosis | n (%) |
| Focal segmental glomerulosclerosis (FSGS) | 78 (26.0) |
| Lupus nephritis | 63 (21.0) |
| Membranous nephropathy (MN) | 45 (15.0) |
| AA amyloidosis | 28 (9.3) |
| Membranoproliferative GN (MPGN) | 23 (7.6) |
| Minimal change disease (MCD) | 17 (5.6) |
| IgA nephropathy | 13 (4.3) |
| Diabetic nephropathy | 9 (3.0) |
| Crescentic GN | 9 (3.0) |
| Multiple myeloma–related kidney disease | 6 (2.0) |
| Chronic nephropathy (global glomerulosclerosis) | 3 (1.0) |
| AL amyloidosis | 4 (1.3) |
| Fabry disease | 2 (0.7) |
Primary vs. secondary nephrotic syndrome
Nephrotic syndrome was classified as primary in 156 patients (50.7%). Secondary causes were mainly lupus nephritis (21%) and renal amyloidosis (AA 9.3%, AL 1.3%). Multiple myeloma accounted for 2%, diabetes for 3%, and Fabry disease for 0.7%.
Patients without kidney biopsy (n = 8; 2.5%)
Kidney biopsy was not performed due to contraindications, mainly coagulation disorders and renal structural abnormalities (renal atrophy, congenital anomalies, renal ectopia), as well as specific situations such as microaneurysms, solitary kidney, and advanced CKD with small kidneys [8,9].Etiologies among these patients were: AA amyloidosis (n = 2) (supported by clinico-biological findings and confirmed by minor salivary gland biopsy), multiple myeloma (n = 1), secondary membranous nephropathy related to tuberculosis (n = 1), and diabetic nephropathy (n = 4). Diagnoses were based on clinico-biological and/or extra-renal histological evidence when indicated.
Treatment
All patients received symptomatic management [10]:
- Renoprotective therapy (mainly ACE inhibitors): 301 patients (98%)
- Edema management (low-sodium diet + loop diuretics): 271 patients (88%)
- Prophylactic anticoagulation (vitamin K antagonists): 179 patients (58%)
- Hemodialysis: 63 patients (20%)
Outcomes
Complete remission occurred in 54%, and end-stage kidney disease (ESKD) in 17% (Table 4).
| Table 4: Outcomes (N = 308). | |
| Outcome | n (%) |
| Complete remission | 168 (54.0) |
| ESKD | 53 (17.0) |
| Death | 11 (3.5) |
Complications
Nephrotic syndrome–related complications occurred in 131 patients (43.4%). Acute kidney injury (AKI) was the most frequent complication.
Complicated vs. non-complicated patients
Among patients with complications, underlying histology most frequently included lupus nephritis (n = 48), membranous nephropathy (MN) (n = 25), and amyloidosis (n = 12) (Table 5).
| Table 5: Baseline characteristics according to complications status. | ||
| Characteristic | With complications (N = 131; 43%) | Without complications (N = 177; 57%) |
| Male-to-female ratio | 0.8 | 1.03 |
| Age (years), mean ± SD | 46 ± 15 | 40 ± 17 |
| Urine output (L/24 h) | 1.65 | 1.76 |
| Serum creatinine at baseline (mg/L), median | 54 | 17 |
| Serum albumin at baseline (g/L), median | 19 | 21 |
| 24-h proteinuria at baseline (g/24 h), median | 7.9 | 6.6 |
| Primary vs. secondary glomerulopathy, n (%) | 55 (41) / 76 (58) | 97 (55) / 80 (45) |
Renal complications
Renal complications were common; acute kidney injury (AKI) occurred in 111 patients (36% of the overall cohort). Among patients with AKI, 66 progressed to chronic kidney disease (CKD), whereas 45 recovered renal function with normalization of serum creatinine after treatment.
Mean urine output was lower in AKI (1.31 L/24 h) compared with CKD (1.82 L/24 h). Management included optimization of hemodynamics (including fluid management as indicated), discontinuation of nephrotoxic drugs, and initiation of dialysis in selected patients with advanced CKD.
Infectious complications
Infectious complications occurred in 37 patients (12% of complicated patients). Reported infections included spontaneous bacterial peritonitis (n = 6; 1.9%), urinary tract infections (n = 18; 5.8%), and pulmonary infections (n = 13; 4.2%). On urinalysis/culture, leukocyturia was found in 34% and hematuria in 22% of infected patients. Patients were treated with targeted antibiotic therapy.
Thromboembolic complications
Thromboembolic events occurred in 11 patients (3.5%), including deep vein thrombosis (DVT) and pulmonary embolism (PE). Nine patients developed lower-limb femoral/popliteal DVT (one associated with PE), and two patients had PE.
Metabolic and endocrine complications
After etiologic treatment, 5 patients (1.7%) had persistent moderate dyslipidemia. Steroid-induced diabetes occurred in 29 patients (9.6%), and hypothyroidism in 14 patients (5.2%) (Table 6).
| Table 6: Characteristics of complications in patients with nephrotic syndrome. | |||||||
| Variable (baseline, M0) | AKI (n = 111; 36%) | CKD (n = 66; 21%) | DVT/PE (n = 12; 3.8%) | Infections (n = 37; 10%) | Steroid-induced diabetes (n = 29; 9%) | Dyslipidemia (n = 5; 1.6%) | Hypothyroidism (n = 16; 5%) |
| Age (years), mean ± SD | 43 ± 14.2 | 47 ± 17 | 38 ± 18 | 42.5 ± 15.6 | 50 ± 12.9 | 53 ± 19 | 39 ± 15.6 |
| Male-to-female ratio | 0.6 | 0.8 | 0.8 | 0.6 | 0.7 | 2.0 | 0.5 |
| Weight (kg) | 80 | 73 | 59 | 88 | 80 | 81 | 71 |
| Urine output (L/24 h) | 1.31 | 1.82 | 1.40 | 1.60 | 1.35 | 1.40 | 1.55 |
| Serum creatinine (mg/L) | 38 | 66.4 | 14.9 | 100 | 21 | 18 | 12.5 |
| Total serum protein (g/L) | 49 | 49 | 45 | 49 | 48 | 45 | 57 |
| Serum albumin (g/L) | 19 | 20 | 17 | 16 | 23 | 14 | 26 |
| Fasting plasma glucose (g/L) | 0.78 | 0.94 | 0.94 | 0.78 | 0.85 | 0.82 | 0.81 |
| Total cholesterol (g/L) | 2.40 | 2.74 | 3.60 | 2.40 | 2.95 | 3.55 | 1.70 |
| HDL cholesterol (g/L) | 0.33 | 0.41 | 0.33 | 0.40 | 0.42 | 0.49 | 0.47 |
| LDL cholesterol (g/L) | 1.70 | 2.05 | 2.16 | 1.60 | 2.10 | 2.73 | 1.20 |
| Triglycerides (g/L) | 1.48 | 2.05 | 2.68 | 2.00 | 2.00 | 3.07 | 1.31 |
| TSH (UI) | 4.50 | 7.10 | 2.50 | 4.70 | 3.22 | 4.46 | 1.42 |
| Urine culture/urinalysis—leukocyturia (ECBU-L) | 14 | 25.3 | 29 | 14 | 19 | 35 | 32 |
| Urine culture/urinalysis—hematuria (ECBU-H) | 12 | 70 | 17 | 12 | 11 | 14 | 33 |
| 24-h proteinuria (g/24 h) | 9.0 | 8.37 | 8.90 | 9.0 | 5.0 | 8.5 | 5.0 |
| Notes: AKI = Acute Kidney Injury; CKD = Chronic Kidney Disease; DVT/PE = Deep Vein Thrombosis / Pulmonary Embolism; TSH = Thyroid-Stimulating Hormone. | |||||||
Risk factor analysis for complications (CPC)
Risk factors associated with the occurrence of complications (CPC) were assessed using logistic regression in univariable and multivariable analyses (Table 7).
| Table 7: Risk factors associated with complications (CPC): univariable and multivariable logistic regression. | ||||||
| Risk factor | Univariable OR | 95% CI | p | Multivariable OR | 95% CI | p |
| Age | 1.02 | 1.013–1.043 | <0.001 | 1.028 | 0.98–1.08 | 0.268 |
| Sex | 1.174 | 0.399–3.450 | 0.977 | 9.881 | 0.277–352.491 | 0.977 |
| Hypertension | 0.488 | 0.285–0.835 | 0.033 | 12.155 | 0.933–158.300 | 0.056 |
| Smoking | 0.791 | 0.421–1.484 | 0.487 | 0.652 | 0.054–7.863 | 0.736 |
| Weight | 1.034 | 1.016–1.052 | <0.001 | 1.083 | 1.010–1.162 | 0.024 |
| Serum creatinine at admission | 1.030 | 1.021–1.040 | <0.001 | 1.040 | 1.014–1.068 | 0.003 |
| Total serum protein | 1.011 | 0.985–1.038 | 0.392 | 0.991 | 0.896–1.095 | 0.853 |
| Serum albumin | 0.951 | 0.909–0.995 | 0.028 | 0.874 | 0.710–1.077 | 0.207 |
| 24-hour proteinuria | 1.119 | 1.045–1.199 | 0.001 | 1.228 | 0.991–1.520 | 0.060 |
In univariable analysis, age (p < 0.001), hypertension (p = 0.033), weight (p < 0.001), serum creatinine at admission (p < 0.001), serum albumin (p = 0.028), and 24-hour proteinuria (p = 0.001) were significantly associated with CPC.
In multivariable analysis, only higher weight (p = 0.024) and serum creatinine at admission (p = 0.003) remained independently associated with CPC.
Risk factors for progression to chronic kidney disease (CKD)
After adjustment for age, sex, diabetes, hypertension, smoking, edema, weight, urine output, total serum protein, serum albumin, urinalysis/culture parameters (leukocyturia and hematuria), and 24-hour proteinuria, the only factor independently associated with progression to CKD was serum creatinine (p = 0.001) (Table 8).
| Table 8: Risk factors for progression to CKD: univariable and multivariable logistic regression. | ||||||
| Risk factor | Univariable OR | 95% CI | p | Multivariable OR | 95% CI | p |
| Age | 1.026 | 1.010–1.042 | 0.001 | 1.050 | 0.993–1.110 | 0.086 |
| Sex | 0.533 | 0.179–1.586 | 0.153 | 1.003 | 0.026–39.448 | 0.998 |
| Edema syndrome | 1.202 | 0.525–2.754 | 0.663 | 0.269 | 0.015–4.669 | 0.367 |
| Hypertension | 0.345 | 0.199–0.597 | 0.001 | 0.082 | 0.007–1.384 | 0.088 |
| Weight at baseline (M0) | 1.023 | 1.005–1.041 | 0.012 | 0.983 | 0.894–1.082 | 0.730 |
| Urine output | 1.180 | 0.925–1.504 | 0.182 | 1.579 | 0.349–7.147 | 0.553 |
| Serum creatinine | 1.038 | 1.028–1.048 | <0.001 | 1.051 | 1.021–1.082 | 0.001 |
| Total serum protein at baseline (M0) | 0.984 | 0.957–1.013 | 0.279 | 1.018 | 0.909–1.140 | 0.761 |
| Serum albumin at baseline (M0) | 0.966 | 0.922–1.013 | 0.156 | 0.823 | 0.633–1.071 | 0.147 |
| Leukocyturia at baseline (ECBU-L, M0) | 0.998 | 0.993–1.003 | 0.484 | 0.992 | 0.979–1.004 | 0.188 |
| Hematuria at baseline (ECBU-H, M0) | 1.002 | 1.000–1.004 | 0.066 | 0.999 | 0.982–1.017 | 0.903 |
| 24-hour proteinuria at baseline (M0) | 1.144 | 1.066–1.227 | <0.001 | 1.219 | 0.950–1.565 | 0.120 |
Age showed a non-significant trend (p = 0.086), while 24-hour proteinuria was not significantly associated after adjustment (p = 0.120).
Key takeaways
- For overall complications (CPC), baseline weight and admission creatinine were the only independent predictors in the multivariable model.
- For progression to CKD, serum creatinine was the sole independent predictor after full adjustment.
Nephrotic syndrome (NS) shows substantial epidemiological variation across regions and study populations, influenced by age, sex distribution, access to kidney biopsy, and the local burden of systemic diseases. In our cohort, the mean age was 42.5 ± 15 years, reflecting a wide age range and a predominantly middle-aged adult population. This is lower than the mean age reported by Cheikh Diouf, et al. [6] in Senegal (65.7 ± 6.2 years), highlighting potential differences in referral patterns, comorbidity profiles, or inclusion criteria across settings [11].
Sex distribution in our cohort was nearly balanced (51% men, 49% women), contrasting with multiple reports describing male predominance in NS. For instance, a study from Mali reported a male-to-female ratio of 2.22. Such discrepancies may reflect etiological heterogeneity, as some NS entities are more frequent in men, whereas systemic lupus erythematosus (SLE)—a major secondary cause—predominantly affects women. Our findings, therefore, likely capture a mixed etiological spectrum in which female-predominant causes (e.g., lupus nephritis) counterbalance male-predominant patterns reported elsewhere.
Primary vs. secondary glomerular diseases
Etiologies of NS can be broadly categorized into primary and secondary glomerulopathies. In our study, primary glomerulopathies predominated, with focal segmental glomerulosclerosis (FSGS) as the most frequent primary diagnosis. This aligns with an African meta-analysis reporting FSGS as the leading primary lesion (~26%), followed by minimal change disease and membranous nephropathy (MN). In contrast, in Europe and North America, MN is often the predominant cause of adult NS in non-diabetic Caucasian populations, with an estimated annual incidence of 1.2–1.7 per 100,000 adults, underscoring important geographic differences.
Secondary glomerulopathies represented a substantial proportion of NS in our cohort, mainly driven by lupus nephritis, diabetic kidney disease, and amyloidosis. This is consistent with the growing impact of cardiometabolic and systemic inflammatory diseases in many African settings. Lupus nephritis remains a key cause of glomerular disease among younger adults, especially women, while AA amyloidosis is classically associated with chronic inflammatory conditions [12].
Kidney outcomes and renal complications
Chronic kidney disease (CKD) is a major complication of NS. In our cohort, 21% of patients progressed to CKD. This finding is consistent with previous studies reporting a significant long-term risk of CKD in certain primary lesions such as FSGS. The risk may be even higher in secondary forms, including amyloidosis and diabetic kidney disease, where progression can be accelerated by delayed diagnosis or limited disease-specific therapeutic options.
In addition, acute kidney injury (AKI) occurred in 36% of the overall cohort, emphasizing the importance of early recognition and management of precipitating factors such as intravascular volume depletion, nephrotoxic exposures, infections, and hemodynamic instability. In clinical practice, optimizing supportive care—including renin–angiotensin system blockade when appropriate, edema control, and individualized fluid management—may contribute to renal recovery in some patients.
Dyslipidemia and metabolic/endocrine complications
Dyslipidemia is a well-recognized feature of NS, typically involving increased total cholesterol, LDL cholesterol, and triglycerides, with reduced HDL cholesterol, thereby contributing to elevated cardiovascular risk. Our data confirm that lipid abnormalities are frequent at baseline in NS, consistent with reports indicating that dyslipidemia affects approximately 60% – 80% of patients [13].
However, in our cohort, persistent moderate dyslipidemia after etiologic treatment remained relatively uncommon, affecting only a small proportion of patients (1.6% – 1.7%). These findings suggest that although dyslipidemia is frequent at presentation, it may improve with appropriate treatment of the underlying nephrotic syndrome.
Steroid-induced diabetes was also observed, reflecting the metabolic burden associated with immunosuppressive therapies used in NS. Thyroid dysfunction, particularly hypothyroidism, may also occur due to urinary loss of thyroid hormone–binding proteins and therefore requires monitoring in patients with prolonged or severe NS [14].
Thromboembolic and infectious complications
NS is associated with an increased risk of thromboembolic events through multiple mechanisms, including urinary losses of anticoagulant proteins such as antithrombin, increased hepatic synthesis of procoagulant factors, platelet hyperreactivity, and hemoconcentration. Hypoalbuminemia is frequently used as a risk stratifier when considering thromboprophylaxis.
In our cohort, thromboembolic events occurred in a small proportion of patients, supporting the need for individualized prophylactic strategies based on both thrombotic and bleeding risks.
Infectious complications were observed in 10% of patients, a rate slightly lower than that reported in some series. This variability may reflect differences in exposure to immunosuppressive therapy, vaccination practices, diagnostic intensity, or case-mix across studies. Reported infections included spontaneous bacterial peritonitis and respiratory infections, highlighting the importance of early recognition, appropriate antimicrobial therapy, and preventive measures such as vaccination and patient education.
Predictors of complications and progression
Our regression analyses identified baseline weight and admission serum creatinine as independent predictors of complications. One possible explanation is that increased body weight may reflect fluid retention and more severe edema in nephrotic syndrome, which may in turn indicate a higher disease burden and greater risk of complications. In addition, excess body weight may contribute to systemic inflammation and metabolic disturbances that can worsen renal outcomes [15-19].
Regarding progression to CKD, serum creatinine remained the only variable independently associated after adjustment, while age showed a non-significant trend. These findings emphasize that patients presenting with higher creatinine levels represent a higher-risk group and may benefit from closer monitoring and early optimization of both supportive and disease-specific therapies [20-23].
This study highlights the clinical burden of nephrotic syndrome in an African adult cohort, characterized by a predominance of FSGS among primary glomerulopathies and a substantial contribution of secondary causes, notably lupus nephritis and diabetic kidney disease. Renal complications were frequent, including AKI and progression to CKD (21%), with baseline serum creatinine emerging as a key independent predictor of adverse renal evolution. Thromboembolic and infectious events, although less frequent than in some reports, remain clinically meaningful and support early risk stratification and targeted preventive strategies. Overall, these findings underscore the need for timely diagnosis, careful supportive management, and coordinated multidisciplinary care to reduce morbidity and slow renal progression in high-risk patients.
- Niaudet P. Nephrotic syndrome in children. In: Encycl Med Chir. Nephrology-Urology; Pediatrics. Paris: Elsevier SAS; 2000. Issue 18-039-D10:12.
- Kidney Disease: Improving Global Outcomes (KDIGO) Glomerulonephritis Work Group. KDIGO clinical practice guideline for glomerulonephritis. Kidney Int Suppl. 2012;2(2):139-274.
- Seggie JL. Nephrotic syndrome in tropical Africa: glomerulonephritis in Zimbabwe. In: Nephrology, kidney diseases in the tropics. Oxford: Oxford University Press; 2006;1030-1040.
- Anigilaje EA. Childhood nephrotic syndrome at the University of Abuja Teaching Hospital, Abuja, Nigeria: a preliminary report supports high steroid responsiveness. Sudan J Paediatr. 2019;19(2):126-139. Available from: https://doi.org/10.24911/sjp.106-1547399573
- Balde MS. Idiopathic nephrotic syndrome in the pediatric department of the Gabriel Toure University Hospital. Doctoral thesis. Bamako: University of Sciences, Techniques and Technologies of Bamako; 2019.
- Diouf BA. Nephrotic syndrome in the elderly in Senegal: epidemiological, etiological, therapeutic, and prognostic profiles. Nephrol Ther. 2017;13(6):446-451.
- Hogan JJ, Mocanu M, Berns JS. The native kidney biopsy: update and evidence for best practice. Clin J Am Soc Nephrol. 2016;11(2):354-362. Available from: https://doi.org/10.2215/cjn.05750515
- Bandari J, Fuller TW. Renal biopsy for medical renal disease: indications and contraindications. Can J Urol. 2016;23(1):8121-8126. Available from: https://pubmed.ncbi.nlm.nih.gov/26892051/
- Bourial MG. Epidemiology of glomerulonephritis and indications for renal biopsy in children: 10 years of experience. Nephrol Ther. 2017;13(5):395-396.
- Crew RJ. Complications of the nephrotic syndrome and their treatment. Clin Nephrol. 2004;62(4):245-259. Available from: https://doi.org/10.5414/cnp62245
- Glassock RJ. Prophylactic anticoagulation in nephrotic syndrome: a clinical conundrum. J Am Soc Nephrol. 2007;30(7):1123-1125. Available from: https://doi.org/10.1681/asn.2006111300
- Almaani S, Meara A, Rovin BH. Update on lupus nephritis. Clin J Am Soc Nephrol. 2017;12(5):825-835. Available from: https://doi.org/10.2215/cjn.05780616
- Hari P, Khandelwal P, Smoyer WE. Dyslipidemia and cardiovascular health in nephrotic syndrome. Pediatr Nephrol. 2019;34(4):813-824. Available from: https://doi.org/10.1007/s00467-019-04301-y
- Sinha A, Bagga A. Steroid-sensitive nephrotic syndrome: revised guidelines. Indian Pediatr. 2021;58(5):461-481. Available from: https://www.indianpediatrics.net/may2021/may-461-481.htm
- Karakaya D, Gungor T, Kargin Cakici E, Yazilitas F, Celikkaya E, Bulbul M. Complications related to idiopathic nephrotic syndrome, its treatment, and the associated risks in patients. Cureus. 2023;15(8):e43929. Available from: https://doi.org/10.7759/cureus.43929
- Kay J. Venous thromboembolism prevention in nephrotic syndrome: the case for aspirin. Kidney Int Rep. 2025;10(2):123-130.
- Keita Y, Leye A. Le syndrome nephrotique idiopathique de l’enfant a Dakar: a propos de 40 cas. Pan Afr Med J. 2017;26:106.
- Korbet SM. Treatment of primary focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2002;7(4):448-456.
- Musunuru K, Kathiresan S. Dyslipidaemia in nephrotic syndrome: mechanisms and treatment. Nat Rev Nephrol. 2018;14(1):57-70.
- Ponticelli C, Glassock RJ. Current and emerging treatments for idiopathic focal and segmental glomerulosclerosis in adults. Expert Rev Clin Immunol. 2013;9(6):629-637.
- Toure YI. The role of nephrology in the morbidity of an internal medicine ward for Black African adults in Dakar: a study of 7379 cases. Dakar Med. 1984;29:213-220.
- Floege J. Primary glomerulonephritides. Lancet. 2016;393(10174):1642-1656.
- Gigante A, Baiocchi B. Nephrotic syndrome and stroke. Int J Immunopathol Pharmacol. 2013;26(3):769-772. Available from: https://doi.org/10.1177/039463201302600322